4 min read
News: Alcami and UNCW Awarded Biomanufacturing Workforce Development Grant
Project Call 1.0 Proposal Selected by NIIMBL Governing Committee
Alcami and the University of North Carolina Wilmington (UNCW) have partnered on an academic program that connects education and work experiences for students pursuing scientific careers, especially in pharmaceutical sciences. This edition of the Beyond the Bench series will highlight the laboratory-based course taught at UNCW’s state-of-the-art marine biotechnology center (MARBIONC), “Current Good Pharmaceutical Quality Control Laboratory Practice,” focused on the application of Current Good Manufacturing Practices (cGMPs) in drug development and testing laboratories.
In 2016, leaders from Alcami and UNCW met to discuss potential opportunities to work together on biopharmaceutical projects. Many of Alcami’s Wilmington employees are UNCW graduates, and Alcami has held a long history of commitment to the university on employee education programs and pharmaceutical projects. Steve Fontana, JD, from UNCW’s Office of Intellectual Property and Commercialization; UNCW Chancellor Dr. Jose V. Sartarelli, and Alcami leaders initiated conversations that resulted in the development of the present partnership.
“Alcami professionals have the expertise to prepare students for careers in the biopharmaceutical industry, and the executive leadership of Alcami and UNCW developed a plan to offer courses to students. Both sides are benefitting from this partnership,” said Jonna Clark, Alcami Scientific Advisor and liaison for university relations. “UNCW graduates employed at Alcami have a broader understanding of the pharmaceutical industry that will accelerate their careers. These employees will start their careers understanding the importance of regulatory compliance, safety, quality, honesty, and integrity in the industry.”
Led by Clark, several guest Alcami scientists lectured in the classroom and taught in the labs to provide a robust educational experience. “The UNCW students received hands-on experience within a GMP environment that potential employers will look for in their new hires,” commented Dr. Will Boomershine, a subject matter expert at Alcami for structural chemistry and biotechnology.
Throughout the course, Alcami scientists have shared their experiences with the next generation of scientists who will be the future of Alcami. “I would highly recommend this course to any student interested in a science-related career, not just those interested in pharmaceuticals. Experience in a GMP environment provides a preview of what is expected of a scientist in the pharmaceutical field from both a scientific and regulatory perspective. Even for students who are not interested in pharmaceuticals, the class provides insight into the highly-regulated environment of pharmaceuticals, providing a glimpse into this industry. Having an understanding of GMP regulations can also help academic labs in the development of new drugs,” stated Boomershine.
Kyle Chenevert was a biology and chemistry senior when he took this class and “felt this course prepared me for my career in science by simultaneously giving me experience with GMP lab testing and documentation and helping me build relationships with experienced industry mentors.” Following the course and his graduation from UNCW, Chenevert joined Alcami as an assistant scientist on the structural chemistry and biotechnology team. “In my case, the transition from student to employee was a very comfortable one. Having an opportunity to be exposed to a GMP lab environment prior to my first day of work allowed me to put all of the information I was being given into context. On top of that, the benefit of walking into that first day with a few familiar faces made joining the team a breeze. I’m still learning more with every passing day, but I can say with no hesitation that taking the GMP course at UNCW made a hugely positive impact on my onboarding experience,” Chenevert explained.
Notable favorite topics for the students in the course included dissolutions, bioassay, high-performance liquid chromatography (HPLC), biopharmaceutical bicinchoninic acid (BCA) assay lab, and meeting and networking with a multitude of scientific professionals.
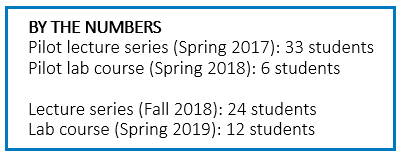
“This class uses real scenarios that we will encounter in the workplace, such as GMP regulations and technique and lab tasks. Most classes merely supply you with a lecture and scenario-based lab, whereas this class uses all of the same materials and instruments that you would find in real life,” said Ashley Bissette, who took the class as a clinical research and biology junior. “I really enjoyed the connections I made throughout this course. It was no secret that this course was a challenge for me from the beginning as I had only taken Chem 101, but Jonna Clark and the other Alcami scientists were always willing to answer questions, help me understand the concepts…[and] apply this knowledge in the lab and see the process behind drug manufacturing,” she said. Bissette went on to intern at Alcami as part of the scientific support services team over the summer.
“As a guest lecturer in the class, the students also taught me many things,” Boomershine said. “First, they shared their interests scientifically and what they are learning in their other classes. They also shared what they are looking for in an employer. As future leaders in the scientific community, it is important to understand what the current students are looking for in an employer so Alcami can attract and retain talented young scientists.”
“I have recommended this class to many students interested in the same path I took, doing lab research work, because it really made a huge difference,” added Bissette. “I feel I have an advantage over my classmates because I have seen cGMP applications first hand. I hope to take the knowledge into my future clinical research career!”

Alcami and UNCW faculty continue to partner closely to offer innovative workforce development solutions for the biopharmaceutical industry. The lecture, laboratory, and internship series offered in 2019 was funded in part by the National Institute for Innovation Manufacturing Biopharmaceuticals (NIIMBL) organization. NIIMBL is funded through a cooperative agreement with The National Institute of Standards and Technology (NIST), a program dedicated to promoting innovation and industrial competitiveness. The ultimate goal is to develop a sustainable program, advance regional expertise, and produce new talent for the pharmaceutical industry.
ABOUT ALCAMI
Alcami is a world-class fully-integrated end-to-end contract development and manufacturing organization (CDMO) headquartered in North Carolina. We provide customizable and innovative services to small and mid-size pharmaceutical and biotechnology companies by offering individualized and integrated services across multiple areas. We connect our clients with innovative solutions for formulation development, analytical development and testing services, clinical and commercial finished dosage form manufacturing (oral solid dose and parenteral), packaging, and stability services. For more information, please visit alcaminow.com.
ABOUT THE UNIVERSITY OF NORTH CAROLINA WILMINGTON
The University of North Carolina Wilmington, the state’s coastal university, is dedicated to learning through the integration of teaching and mentoring with research and service. A public institution with nearly 14,000 students, our university is widely acknowledged for its superb faculty and staff and its powerful academic experience that stimulates creative inquiry, critical thinking, thoughtful expression, and responsible citizenship. With an array of high-quality programs at the baccalaureate and master’s levels, and doctoral programs in marine biology and educational leadership, UNCW is continuously recognized at a national level for academic excellence and affordability. We are dedicated to offering a community rich in diversity and inclusion, global perspectives, and enriching the quality of life through scholarly community engagement in such areas as health, education, the economy, the environment, marine and coastal issues, and the arts. For more information, please visit uncw.edu.
CONTACT UNC WILMINGTON
Janine Iamunno
Chief Communications Officer
601 S. College Road
Wilmington, NC 28403
iamunnoj@uncw.edu
+1 910.962.2445
uncw.edu
ABOUT NIIMBL
NIIMBL is a public-private partnership with the goal of advancing innovation in biopharmaceutical manufacturing. NIIMBL is part of Manufacturing USA®, a network of 14 manufacturing institutes across the country that brings together industry, academia, and the public sector to propel promising research developments, accelerate new products to market, and train tomorrow’s workforce in order to secure America’s future. NIIMBL is funded through a cooperative agreement with the National Institute of Standards and Technology (NIST) in the U.S. Department of Commerce and leverages additional support from industry, academic institutions, non-profit organizations, and the states of Delaware, Maryland, North Carolina, and the Commonwealth of Massachusetts. The NIIMBL mission is to accelerate biopharmaceutical innovation, support the development of standards that enable more efficient and rapid manufacturing capabilities, and educate and train a world-leading biopharmaceutical manufacturing workforce, fundamentally advancing U.S. competitiveness in this industry.
CONTACT NIIMBL
Dan Maiese
NIIMBL Communications Director
dmaiese@udel.edu
302-831-3824
NIIMBL.org
4 min read
Project Call 1.0 Proposal Selected by NIIMBL Governing Committee
5 min read
In recent years 505(b)(2) approvals have experienced double-digit growth. Many companies are electing to pursue 505(b)(2) applications to obtain a...
7 min read
Increasing prevalence of drug misuse and abuse is driving a heightened and more stringent approach to abuse-deterrent formulations. Possibly the most...